

EXPERIMENTAL
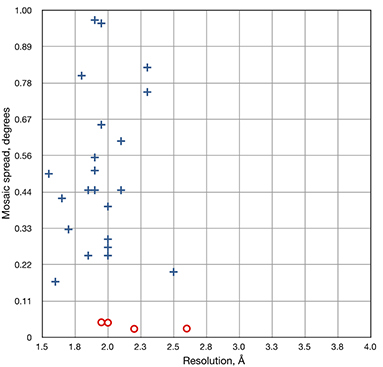
The comparison of mosaic spread for X-CHIP data (shown as red circles) with synchrotron cryo data sets (shown as blue crosses) from unique crystal structures.
The Advantages of X-CHIP in X-ray Data Collection and Crystallographic Refinement
| Data* | T,K | a, Å | b, Å | c, Å | ß,° | d, Å | Rwork | Rfree | |
| Cryo Loop | 100 | 52.9 | 38.3 | 77.0 | 102.4 | 1.9 | 0.970 | 17.6 | 19.6 |
| X-CHIP | 298 | 54.3 | 39.0 | 77.0 | 102.0 | 2.0 | 0.045 | 16.1 | 18.7 |
*Protein: EphA3 kinase, P2(1) spase group; #Δ − Mosaic Spread
Cryo freezing causes an anisotropic shift in crystal unit cell constants. The application of X-CHIP technology leads to significant improvement in mosaic spread of the crystal and noticeable improvement to Rwork and Rfree during crystallographic refinement, yielding the crystal structure of the protein molecule in its most natural state.
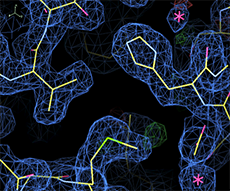 Cryo loop: 1.9 Å, <B>=15 Å2
Cryo loop: 1.9 Å, <B>=15 Å2
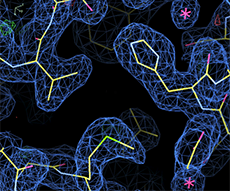 X-CHIP: 2.0 Å, <B>=22 Å2
X-CHIP: 2.0 Å, <B>=22 Å2